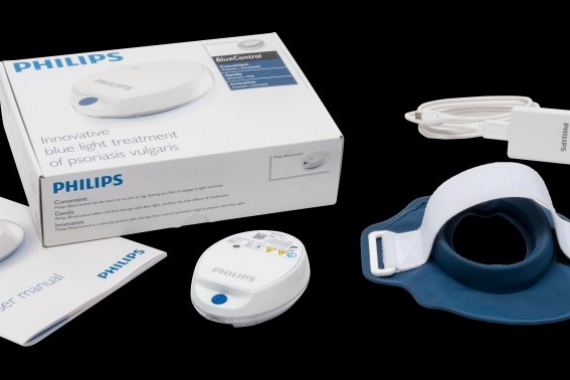
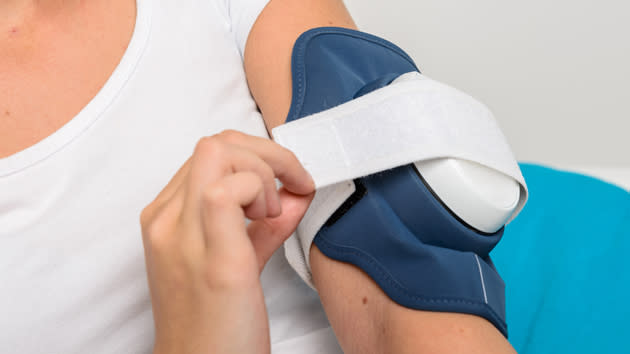
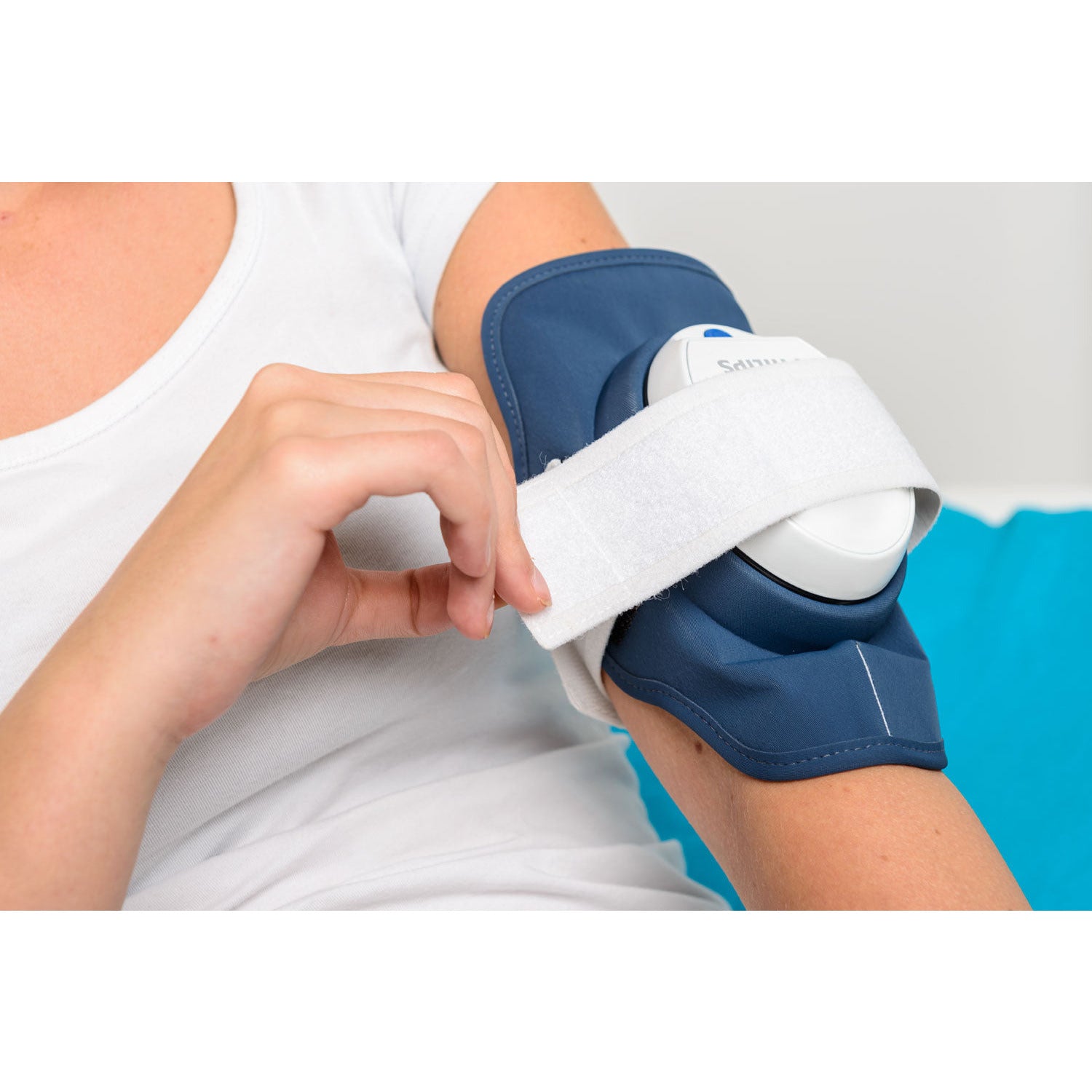
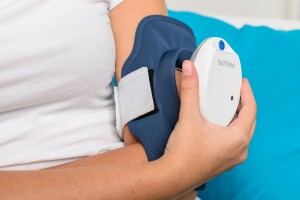
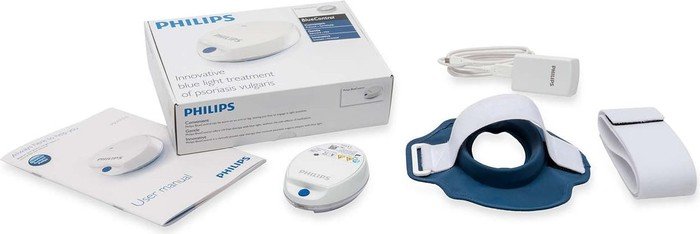
Philips receives U.S. FDA 510(k) clearance to market its Philips BlueControl, a wearable light therapy

BlueLED Therapy on X: "Designed to treat mild-moderate plaque #psoriasis on the arms and legs. Meet Philips #BlueControl. https://t.co/jh77rD99sy https://t.co/DwYVEmvwLO" / X